Advertisement
What are Brownian movement?
Brownian movement. When a colloidal solution is examined with an ultra-microscope, the suspended particles are seen to be in a constant rapid zig-zag motion. This constant rapid zig-zag motion of colloidal particles is known as Brownian movement after the name of the discoverer Sir Robert Brown (1827).
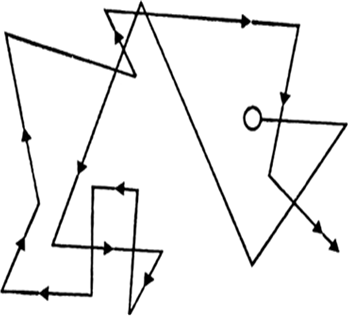
Fig. Brownian movement.
Causes: It is due to the unequal bombardment of colloidal particles by the molecules of the dispersion medium. With increase in the size of the particles, the probabilities of unequal bombardment decreases and the Brownian movement disappears. This is why coarse suspensions do not exhibit Brownian movement.
805 Views
Advertisement
Some zeolites are used to separate long straight chain hydrocarbons from branched chain hydrocarbons in petrochemical industry. Explain how this separation becomes possible.
172 Views
Consider the adsorption isotherm given below and interpret the variation in the extent of adsorption (x/m) when:
(i) temperature increases at constant pressure.
(ii) pressure increases at constant temperature.
403 Views
Advertisement
Advertisement
<
Advertisement