 Multiple Choice Questions
Multiple Choice QuestionsChoose the incorrect statement.
Primary amines show intermolecular hydrogen bonds
Tert-butylamine is a primary amine
Tertiary amines do not show intermolecular hydrogen bonds
Isopropylamine is a secondary amine
Amine that cannot be prepared by Gabriel phthalimide synthesis is
aniline
benzylamine
methylamine
iso-butylamine
Which of the following is the least basic amine?
Ethylamine
Diethylamine
Aniline
Benzylamine
C.
Aniline
Basicity of amines can be explained on the basis of availability of electron density on N-atom for protonation. Because of the + I effect, alkyl group increases the electron density on N-atom in alkyl amines. However, aniline is less basic because phenyl group exerts - I effect, ie, it withdraws electrons
The correct order of increasing basic nature of the following bases is
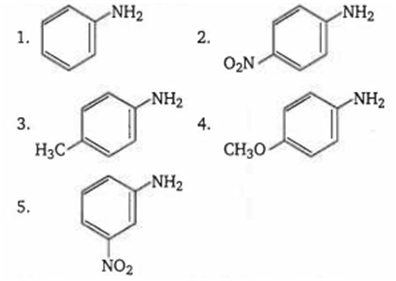
2 < 5 < 1 < 3 < 4
5 < 2 < 1 < 3 < 4
2 < 5 < 1 < 4 < 3
5 < 2 < 1 < 4 < 3
Choose the amide which on reduction with LiAlH4 yields a secondary amine
ethanamide
N-methylethanamide
N, N-dimethylethanamide
phenylmethanamide
Arrange the following amines in the decreasing order of their basic strength. Aniline (I), Benzylamine (II), p-toluidine (III)
I > II > III
II > III > I
III > II > I
II > I > III
Which one of the following compounds will dissolve in an alkali solution after it has undergone reaction with Hinsberg reagent?
CH3NH2
(CH3)3N
(C2H5)2NH
C6H5NHC6H5
Identify the product in the following sequence
3, 4, 5-tribromobenzene
1, 2, 3-tribromobenzene
2, 4, 6-tribromobenzene
3, 4, 5-tribromonitrobenzene
Among the amines
(A) C6H5NH2
(B) CH3NH2
(C) (CH3)3N
(D) (CH3)2NH
A < B < D <C
D < C < B <A
A > B > C >D
B < C < D <A
