 Long Answer Type
Long Answer TypeWhich of the following combinations of orbtials are allowed in LCAO method (considering z-axis to be molecular axis) and sketch the shapes of molecular orbitals formed by their addition and subtraction:
(i) s and pz
(ii) px and px
(iii) pz and py
(iv) s and px.
 Short Answer Type
Short Answer TypeGive the number of electrons which occupy the bonding molecular orbital in H2+ H2 and He2.
What do you understand by bond order? How is it related with bond length and bond energy? Explain on the basis of bond order that He2 molecule does not exist.
Why the bond orders of He2+ and H2– are identical?
Molecular ion Molecular orbital configuration
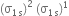

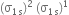
Since both He2+ and H2– have equal number of bonding (Nb) and antibonding (Na) electrons because of identical molecular orbital configuration, their bond order is given by 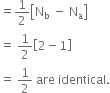
 Long Answer Type
Long Answer TypeUsing LCAO method for the formation of molecular orbitals in case of homonuclear diatomic hydrogen molecule.
Draw the molecular orbital diagram for:
(i) Be2
(ii) B2 and predict bond order and magnetic properties.
 Short Answer Type
Short Answer Type