 Multiple Choice Questions
Multiple Choice QuestionsThe polypeptides are obtained by assembling the peptide units by
ionic bond
covalent bond
intermolecular H-bonding
covalent and H-bonding
Which one of the following have linear structure?
(I) I
(II) NO
(III) I
(IV) SO2
(V) N
I, II and III
I and V
II, III and IV
All of these
The dipole moment of HBr is 1.6 × 10-30 cm and inter atomic spacing is 1 Å. The % ionic character of HBr is
7
10
25
27
The bond order of H If it has 2 bonding electron, how many antibonding electrons it will have?
4
3
2
1
A magnetic moment of 1.73 BM will be shown by which one among the following compounds
[Cu(NH3)4]2+
[Ni(CN)4]2-
TiCl4
[CoCl6]4-
| Iodide | PI3 | AsI3 | SbI3 |
| Bond angle | 102 | 100°2' | 99° |
due to small size of P
due to more bp-bp repulsion in PI3
due to less electronegativity of P
None of the above
The shape of [PtCl3(C2H4)]- and the hybridisation of Pt respectively are
tetrahedral, sp3
trigonal pyramidal, sp3
square planar, dsp2
square planar, d2sp3
C.
square planar, dsp2
Zeise's salt : [K[PtCl3(η2-C3H4)] is a -complex and square planar in structure with dsp2 hybridisation.
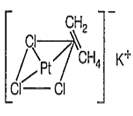
Hybridisation is [PtCl3(η2-C2H4)]-
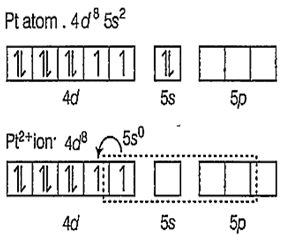
As Cl- ions and C2H4 approaches, the pairing of the unpaired electrons of d-orbitals takes place.

Among the following compounds both coloured and paramagnetic one is
K2Cr2O7
VOSO4
(NH4)2.[TiCl2]
K3[Cu(CN)4]
