 Short Answer Type
Short Answer TypeThe choice of a reducing agent in a particular case depends on thermodynamic factor. How far do you agree with this statement? Support your opinion with two examples.
Thermodynamic factor helps us in choosing a suitable reducing agent for the reduction of particular metal oxide to the metallic state. From , it is evident that metal oxides for which the standard free energy of formation of their oxides is more negative can reduce those metal oxides for which the standard free energy of formation of their respective oxides is less negative. In other words, any metal will reduce the oxides of other metals which lie above it in the Ellingham diagram because the standard free energy change G0 of the combined redox reaction will be negative by an amount equal to the difference in G0 of the two metal oxides. Hence, both Al, Zn can reduce FeO to Fe but cannot reduce Al2O3 to Al and ZnO. Similarly, C can reduce ZnO but not CO.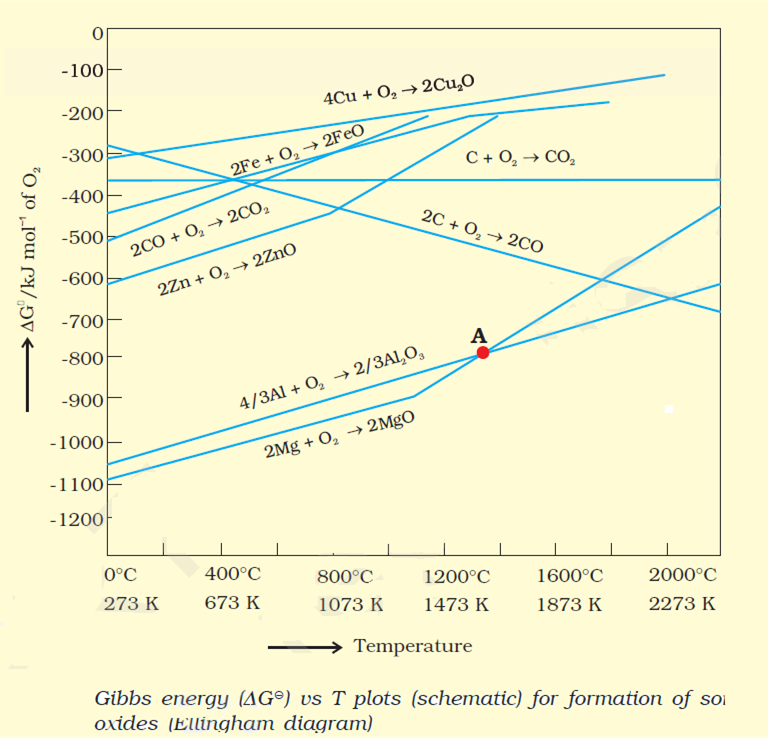
Carbon monoxide is more effective agent that carbon below 983 K but above this temperature the reverse is true. How would you explain this?
The value of ΔrG° for formation of Cr2O3 is – 540 kJmol–1 and that of Al2O3 is – 827 kJ mol-1. Is the reduction of Cr2O3 possible with Al?
Predict the modes of occurrence of the following three types of metals:
(i) Highly reactive (e.g., Na)
(ii) Moderately reactive (e.g., Fe)
(iii) Noble metal (e.g., Au)
 Long Answer Type
Long Answer TypeHow do non-metals occur in nature? How are they extracted/isolated from their natural resources?
 Short Answer Type
Short Answer TypeName the chief ores of tin, iron and aluminium. What methods are employed for the concentration/purification of their ores?
Name three ores which are concentrated by froth-floatation process. What is meant by depressant?
 Long Answer Type
Long Answer TypeDiscuss some of the factors which need consideration before deciding on the method of extraction of metal from its ore.
 Short Answer Type
Short Answer TypeYou are provided with samples of some impure metals such as zinc, copper and germanium. Which methods could you recommend for the purification of each of these metals?
