 Short Answer Type
Short Answer TypeGive a brief account of the following processes as used in extraction of metals:
(a) Smelting
(b) Magnetic separation
Explain how, electrolytic refining purifies a metal?
Electrolytic refining :
In electrolytic refining method, the impure metal is made to act as anode. A strip of the same metal in pure form is used as cathode. They are put in a suitable electrolytic bath containing soluble salt of the same metal. The more basic metal remains in the solution and the less basic go to the anode mud. This can be explained by using electrode potential.
Anode: M → Mn+ + ne-
Cathode : Mn+ + ne- → M
for example copper is refined using an electrolytic method. Anodes are of impure copper and pure copper strips are taken as cathode. The electrolyte is acidified solution of copper sulphate and the net result of electrolysis is the transfer of copper in pure form from the anode to the cathode:
Anode: Cu → Cu2+ + 2 e-
Cathode: Cu2+ + 2 e- → Cu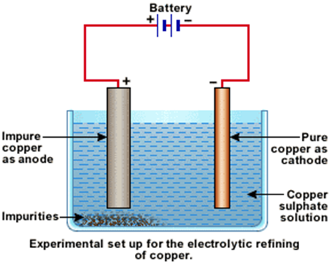
Impurities from the blister copper deposit as anode mud which contains antimony, selenium, tellurium, silver, gold and platinum.
What do you understand by the following terms: (i) Roasting, (ii) Calcination, (iii) Smelting?
 Long Answer Type
Long Answer TypeDescribe mainly with balanced chemical equations.
(a) Copper from copper pyrites by self reduction.
(b) Zinc from zinc blende by carbon reduction.
(c) Silver from silver glance by cyanide process.
(a) How is aluminium extracted from alumina?
(b) Give balanced equations for the extraction of aluminium from bauxite by electrolysis.
(c) What is the function of cryolite in the extraction of Al from Al2O3.
 Short Answer Type
Short Answer Type