 Short Answer Type
Short Answer TypeGive a brief account of the following processes as used in extraction of metals:
(a) Smelting
(b) Magnetic separation
What do you understand by the following terms: (i) Roasting, (ii) Calcination, (iii) Smelting?
 Long Answer Type
Long Answer TypeDescribe mainly with balanced chemical equations.
(a) Copper from copper pyrites by self reduction.
(b) Zinc from zinc blende by carbon reduction.
(c) Silver from silver glance by cyanide process.
(a) How is aluminium extracted from alumina?
(b) Give balanced equations for the extraction of aluminium from bauxite by electrolysis.
(c) What is the function of cryolite in the extraction of Al from Al2O3.
Describe the methods used to obtain concentrated ores.
Method used to obtain concentration:
Hydraulic washing : This method is based on the difference in gravities of the ore and the gangue particles. It is therefore a type of gravity separation. In one such process an upward stream of running water is used to wash the powdered ore. The lighter gangue particles are wasted away and the heavier ores are left behind.
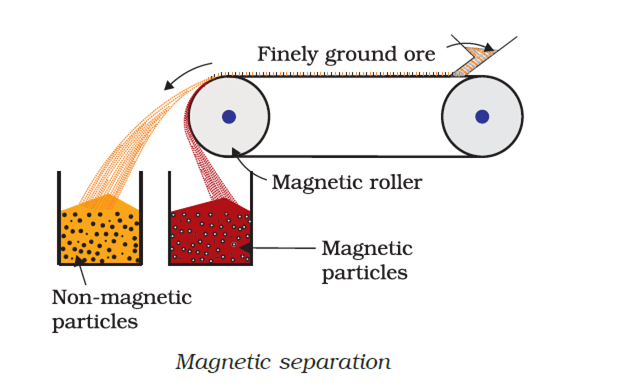
Magnetic separation : this method is based on differences in magnetic properties of the ore components. If either the ore or the gangue is capable of being attracted by a magnetic field, then such separation are carried out. The ground ore is carried on a conveyer belt which passes over a magnetic roller.
Froth floatation method: This method based on removing gangue from sulphide ores. In this process, a suspension of the powdered ore is made with water. To it, collectors and froth stabilisers are added. Collectors (e. g., pine oils, fatty acids, xanthates, etc.) enhance non-wettability of the mineral particles and froth stabilisers (e. g., cresols, aniline) stabilise the froth.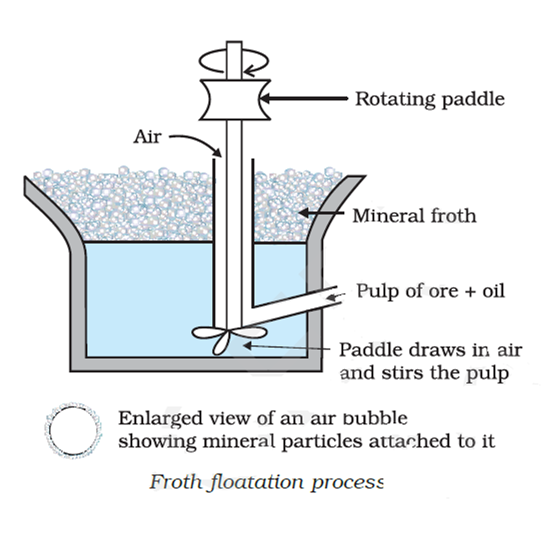
 Short Answer Type
Short Answer Type