 Multiple Choice Questions
Multiple Choice QuestionsAmong the following, the pair in which the two species are not isostructural, is
SiF4 and SF4
IO and XeO3
BH
PF and SF6
The helical structure of protein is stabilized by
dipeptide bond
hydrogen bonds
ether bonds
peptide bonds
The dipole moment of HBr is 1.6 × 10-30 cm and inter-atomic spacing is 1 Å. The % ionic character of HBr is
7
10
15
27
Consider the following ions:
(i) Ni2+ (ii) Co2+ (iii) Cr2+ (iv) Fe3+
(Given, at. no. Cr = 24, Fe = 26, Co = 27, Ni = 28)
The correct order of magnetic moment of these ions is
(i) < (ii) < (iii) < (iv)
(iv) < (ii) < (iii) < (i)
(i) < (iii) < (ii) < (iv)
(iii) < (iv) < (ii) < (i)
Geometry of methyl free radical is
pyramidal
planar
tetrahedral
linear
B.
planar
Number of hybrid orbitals = number of σ bonds + number of lps.
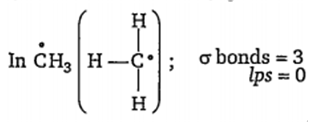
Number of hybrid orbitals =3 + 0 =3
Hence, hybridisation is sp2 and geometry is planar. Methyl free radical is highly reactive due to the presence of one unpaired electron.
