 Multiple Choice Questions
Multiple Choice QuestionsCycloalkane formed when 1,4-dibromopentaneis heated with sodium is
methyl cyclobutane
cyclopentane
cyclobutane
methyl cyclopentane
Least energetic conformation of cyclohexane is
chair conformation
boat conformation
cis conformation
E-Z form
An alkyl bromide (X) reacts with sodium in ether to form 4,5-dIethyl octane, the compound X is
CH3(CH2)3Br
CH3(CH2)5Br
CH3(CH2)3CH(Br)CH3
CH3—(CH2)2—CH(Br)—CH2—CH3
The correct statement is
Cyclohexadene and cyclohexene cannot be isolated with ease during controlled hydrogenation of benzene
One mole each of benzene and hydrogen when reacted gives 1/3 mole of cyclohexane and 2/3 mole unreacted hydrogen
Hydrogenation of benzene to cyclohexane is an endothermic process
It is easier to hydrogenate benzene when compared to cyclohexene
Benzene carbaldehyde is reacted with conc. NaOH solution to give the products A and B. The product A can be used as a food preservative and the product B is an aromatic hydroxy compound, where -OH group is linked to sp3-hybridised C-atom next to benzene ring. The products A and B respectively are
sodium benzoate and phenol
sodium benzoate and phenyl methanol
sodium benzoate and cresol
sodium benzoate and picric acid
The arrangement of following compounds.
(i) Bromomethane
(ii) Bromoform
(iii) Chloromethane
(iv) Dibromomethane
In the increasing order of their boiling point is
III < I < IV < II
IV < III < I < IV
II < III < I < IV
I < II < III < IV
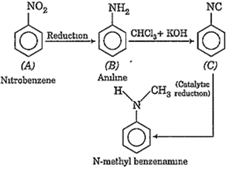
An organic compound A on reduction gives compound B, which on reaction with trichloromethane and caustic potash forms C. The compound C on catalytic reduction gives N-methyl benzenamine, the compound A is,
nitrobenzene
nitromethane
methanamine
benzenamine
A.
nitrobenzene
The compound A is nitrobenzene.
![]() gives aniline on reduction. Thus,
gives aniline on reduction. Thus,
Which of the following has highest knocking ?
Olefins
Branched chain olefins
Straight chain olefins
Aromatic hydrocarbons
Chlorination of benzene is not possible in the following reaction :
C6H6 + Cl2
C6H6 + HOCl
C6H6 + I-Cl
C6H6 + Cl2
