 Multiple Choice Questions
Multiple Choice QuestionsReplacement of Cl of chlorobenzene to give phenol requires drastic conditions, but Cl of 2, 4-dinitrochlorobenzene is readily replaced. This is because
-NO2 group makes the ring electron rich at ortho and para-positions
-NO2 group withdraws electrons from meta-position
-NO2 donates electrons at meta-position
-NO2 withdraws electrons from ortho and para-positions
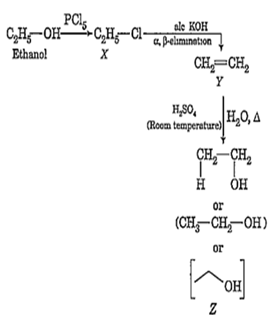
In the reaction,
Ethanol
The product Z is
C2H4
CH3CH2OCH2CH3
CH3CH2OSO3H
![]()
D.
![]()
The product Z is
The reaction which involves dichlorocarbene as an electrophile is
Reimer-Tiemann reaction
Kolbe's reaction
Friedel-Craft's acylation
Fittig reaction
Ethanol is converted into ethoxy ethane,
by heating excess of ethanol with conc. H2SO4 at 140°C
by heating ethanol with excess of conc. H2SO4 at 443 K
by treating with conc. H2SO4 at room temperature
by treating with conc. H2SO4 at 273 K
100 cm2 of 1 M CH3COOH was mixed with 100 cm3 of 2M CH3OH to form an ester. The change in the initial rate if each solution is diluted with equal volume of water would be
2 times
4 times
0.5 times
0.25 times
Arrange the following compounds in the increasing order of their acidic strength.
I. m-nitrophenol
II. m-cresol
III. Phenol
IV. m-chlorophenol
III < II < I < IV
II < IV < III < I
II < III < IV < I
II < III < I < IV
Which of the following functional groups, cannot be reduced to alcohol using NaBH4 in ethanolic solution ?
R - O - R
RCOCl
R - COOH
R - CHO
