 Multiple Choice Questions
Multiple Choice QuestionsReaction of alkyl halides with aromatic compounds in presence of anhydrous AlCl3 is known as:
Friedel-Craft's reaction
Hofmann degradation
Kolbe's synthesis
Beckmann rearrangement
Which of the following will not give iodoform test?
Isopropyl alcohol
Ethanol
Ethanal
Benzyl alcohol
The dehydration of 2-methyl butanol with conc. H2SO4 gives :
2-methyl butene as major product
pentene
2-methyl but-2-ene as major product
2-methyl pent-2-ene
Which of the following order is true regarding the acidic nature of phenol?
Phenol < o-cresol < o-nitrophenol
Phenol > o-cresol > o-nitrophenol
Phenol < o-cresol > o-nitropheno
o-cresol < phenol < o-nitrophenol
Identify the correct statement in the following
n-butane and isobutane are functional isomers
Propan-1-ol and propan-2-ol are position isomers
Dimethyl ether and ethanol are chain isomers
Ethanoic acid and methyl methanoate are position isomerase.
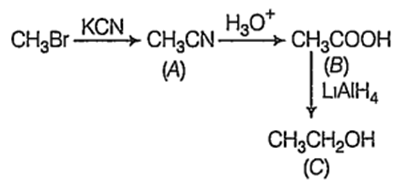
In the following sequence of reactions
The end product C is
acetone
ethyl alcohol
methane
acetaldehyde
B.
ethyl alcohol
Which of the following will not give a positive iodoform test ?
CH3CH2CHOHCH3
CH3CH2CH2COCH3
CH3CH2COCH2CH3
CH3COC6H5
To distinguish between salicylic acid and phenol one can use :
NaHCO3 solution
5% NaOH solution
neutral FeCl3
bromine water
Acid catalyzed hydration of alkenes except ethene leads to the formation of
mixture of secondary and tertiary alcohols
mixture of primary and secondary alcohols
secondary or tertiary alcohol
primary alcohol
Among the following compounds which can be dehydrated very easily ?
CH3CH2CH2CH2CH2OH
CH3CH2CH2CH(OH)CH3
CH3CH2CHCH3
CH3CH2CH(CH3)CH2CH2OH
