 Multiple Choice Questions
Multiple Choice QuestionsPicric acid is :
2, 4, 6-tribromophenol
sym-trinitrophenol
trinitrophenol
2, 4, 6-trinitrotoluene
The cleavage of an aryl-alkyl ether with cold HI gives:
alkyl iodide and water
aryl iodide and water
alkyl iodide, aryl iodide and water
phenol and alkyl iodide
In Lucas test an alcohol reacts immediately and gives insoluble chloride. The alcohol is
CH3OH
CH3CH2OH
(CH3)2CHOH
(CH3)3COH
Glycerol is more viscous than ethanol due to
high molecular weight
high boiling point
many hydrogen bonds per molecule
Fajan's rule
Toluene on treatment with CrO3 and (CH3CO)2O followed by hydrolysis with dil HCl gives
benzaldehyde
benzoic acid
phenol
phenylacetaldehyde
Diacidic base is
CH2(OH)2
Ca(OH)2
CH3CH(OH)2
All of these
B.
Ca(OH)2
Bases are the substance which give -OH ions (hydroxyl ions) when dissolved in
water. If one molecule of a base gives two hydroxyl ions, it is called diacidic base.
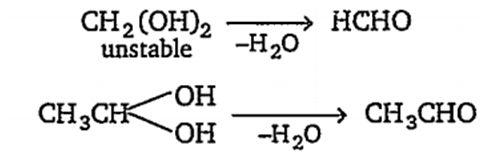
(i) CH2(OH)2 is a covalent compound and C-O bond is more stronger than O-H bond. Hence, it dissociates to give only a less amount of H+ ions instead of OH- ion and acts as acid.

(ii) Ca(OH)2 , being an ionic compound readily gives two hydroxyl ions when dissolved in water, hence it is a diacidic base.
Ca(OH)2 → Ca2+ + 2OH-
(iii) CH3CH(OH)2 is also a covalent compound and gives a small amount of H+ ions, hence it is an acid.

Actually when two-OH groups are attached with same carbon atoms, the compound is highly unstable and loses a water molecule, eg,
Phenol on treatment with diethyl sulphate in presence of NaOH give
phenetole
anisole
diphenyl ether
diethyl ether
