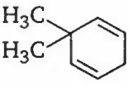Multiple Choice Questions
Multiple Choice QuestionsFructose reduces Tollen's reagent due to
asymmetric carbons
primary alcoholic group
secondary alcoholic group
enolisation of fructose followed by conversion to aldehyde by base
The order of reactivity of phenyl magnesium bromide (PhMgBr) with the following compounds

III > II > I
II > I > III
I > III > II
I > II > III
Match the compounds given in List I with List II and select the suitable option using the codes given below.
| List I | List II | ||
| A. | Benzaldehyde | 1. | Phenolphthalein |
| B. | Phthalic anhydride | 2. | Benzoin condensation |
| C. | Phenyl benzoate | 3. | Oil of wintergreen |
| D. | Methyl salicylate | 4. | Fries rearrangement |
A B C D
4 1 3 2
A B C D
4 2 3 1
A B C D
2 3 4 1
A B C D
2 1 4 3
D.
A B C D
2 1 4 3
(a) Benzoin condensation when an ethanolic solution of benzaldehyde is heated with strong alkali like KCN or NaCN, we get benzoin.

(b)Formation of phenolphthalein: When phenol is treated with phthalic anhydride in the presence of conc. H2SO4 it gives phenolphthalein, an indicator.

(c) Fries rearrangement: Phenyl benzoate when heated with anhydrous AlCl3 in the presence of inert solvent gives ortho-and para-hydroxybenzophenone. In this rearrangement, there is a benzoyl group migration from the phenolic oxygen to an ortho-and para-position.

(d) Methylsalicylate

(A chief constituent of oil of wintergreen)
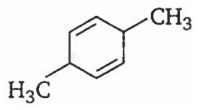
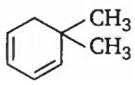
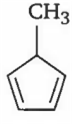
An alkene having molecular formula C8H12 on ozonolysis yields glyoxal and 2, 2-dimethyl butane-l, 4-dial. The structure of alkene is