 Multiple Choice Questions
Multiple Choice QuestionsWhich one of the following will most readily be dehydrated in acidic medium?
CH3—CO—CH2—CH(OH)—CH3
CH3—CHOH—CH2—CH2—CH3
CH3—COCHOH—CH2—CH3
CH3—CO—CH2—CH2—CHOH—CH3
A.
CH3—CO—CH2—CH(OH)—CH3
Greater the conjugation, greater the stability and hence, easier the dehydration. Thus, compound (a), in acidic conditions, will most readily be dehydrated.
In the reaction, CHCl3 + 4OH- → HCOO- + 3Cl- + 2H2O the intermediate species formed is/ are
CCl2
CCl
Both (a) and (b)
None of the above
Consider the following compounds
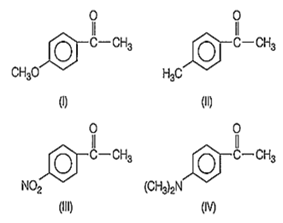
Friedel-Crafts acylation can be used to obtain:
II, III, IV
I, II, IV
I, II, III
I, III, IV
