 Multiple Choice Questions
Multiple Choice QuestionsMark the correct statement.
Methylamine is slightly acidic
Methylamine is less basic than NH3
Methylamine is stronger base than NH3
Methylamine forms salt with alkalies
In the carbylamine reaction, R-X is converted to R-Y via the intermediate Z. R-X, R-Y and Z, respectively, are
RNH2, RNC, Carbene
RNH2, RNC, nitrene
RNC, RNH2, carbene
ROH, RNC, nitrene
Which one of the following is the strongest base ?
2,4,6-trinitroaniline
2,4,6-trinitro-N,N-dimethyl aniline
N, N-dimethyl aniline
Anline
Hinsberg's reagent is
benzene sulphonyl chloride
benzene sulphonic acid
phenyl isocyanide
benzene sulphonamide
The given reaction is called as

Schmidt rearrangement
Curtius rearrangement
Hofmann rearrangement
Lossen rearrangement
Correct basicity of the following compounds are
I. Aniline
II. Pyridine
III. Pyrrole
IV. Guanidine
I >II >III >IV
III >I >II >IV
IV >II >I >III
II >IV>III >I
C.
IV >II >I >III
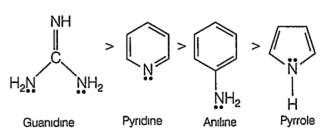
From the structures of the four compounds, It is clear that in pyrrole, the electron pair on nitrogen is involved in delocalisation of the ring. So, it is least basic.
In aniline, the electron pair involved in conjugation with the Π- electrons of the ring while in pyridine it is relatively free. Guandine is a strong base.
One amine is more basic than ammonia and the other is less basic than ammonia. The two amines are respectively
N-methyl ethanamine and N, N-dimethyl ethanamine
aniline and N-methyl aniline
N-methyl aniline and aniline
N, N-dimethyl aniline and benzenamine
One of the isomer of C4H11N is optically active. It must be a
primary amine
secondary amine
tertiary amine
all isomers are optically inactive
Which of the following compounds cannot be identified by carbylamine test?
CHCl3
C6H5-NH-C6H5
C6H5NH2
CH3CH2NH2
