 Short Answer Type
Short Answer TypeWhat is spectrochemical series? Explain the difference between a weak field ligand and a strong field ligand.
Fluorine ion is a weak ligand. It cannot cause the pairing of the 3d electrons. As a result, the Co3+ ion will undergo sp3d2 hybridzation.
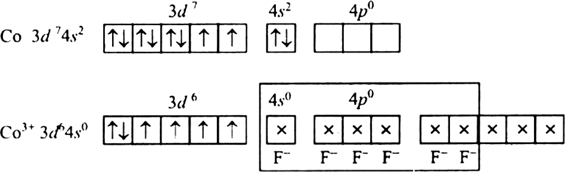
Hence, the geometry of the complex is octahedral and paramagnetic.
