Short Answer Type
Short Answer TypeGive reason for the following: Although aluminium is above hydrogen in the electro-chemical series, it is stable in air and water.
An ore example of galena (PbS) is contaminated with zinc blende (ZnS). Name one chemical which can be used to concentrate galena selectively by froth floatation method.
Why reduction of a metal oxide becomes easier if the metal formed is in liquid state at the temperature of reduction?
If at any site, low grade copper ores are available and zinc and iron scraps are also available. Which of the two scraps would be more suitable for reducing the leached copper ore and why?
Why is the extraction of copper from pyrites difficult than from its oxide ore through reduction.
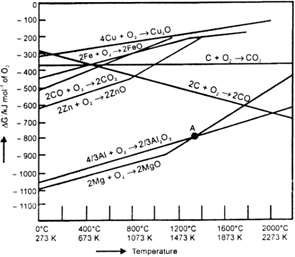
In the graph of ΔrG° Vs T for the formation of oxides, the Cu2O line is almost at the top. So it is quite easier to reduce oxide ores of copper directly to the metal by heating with coke (both the lines of C, CO and C, CO2 are at much lower positions in the graph particularly after 500-600 K).
However sulphide ores are roasted/smelted to give oxides.
2Cu2S + 3O2 → 2Cu2O + 2SO2
The oxide can then be easily reduced to metallic copper using coke.
Cu2O + C → 2Cu + CO