 Multiple Choice Questions
Multiple Choice QuestionsThe process in which metal surface is made inactive is called
passivation
galvanising
corrosion
pickling
Which among the following alloys is used in making instruments for electrical measurements?
Stainless steel
Manganin
Spiegeleisen
Duralumin
Name the metal that is purified by placing the impure metal on sloping hearth of a reverberatory furnace and heating that above its melting point in the absence of air.
Mercury
Gallium
Zirconium
Copper
Name the reagent that is used in leaching of gold.
Carbon
Sodium cyanide
Carbon monoxide
Iodine
Name the process that is employed to refine aluminium.
Hall's process
Mond process
Hoope's process
Serperck's process
C.
Hoope's process
Aluminium is refined by Hoope's process. It consists of carbon electrodes, which are used as cathode and iron tank linked with carbon bricks as anode. The electrolyte consists of three layers-
(i) Bottom layer is made by using impure aluminium
(ii) A molten mixture of fluorides of sodium (Na3AlF6), Ba and Al and Al2O3 is used to
make middle layer.
(iii) At the top, there is a layer of pure molten aluminium.
When electric current is passed, pure aluminium metal is deposited at cathode from the middle layer and an equivalent amount of Al moves from bottom layer to middle layer leaving behind the impurities.
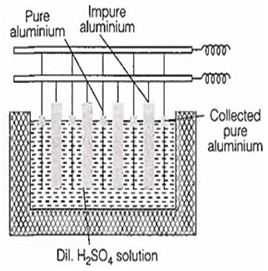
Purification of aluminium by electrolytic refining is called
Hall's process
Froth flotation process
Bayer's process
Hoop's process
