 Multiple Choice Questions
Multiple Choice Questionswhich of the following does not show optical isomerism? (en = ethylenediamine)
[Co(en)2Cl2]+
[Co(NH3)3Cl3]0
[Co(en)Cl2(NH3)2]+
[Co(en)Cl2(NH3)2]+
which of the following complex ions is expected to absorb visible light?
(At. no.Zn = 30, Sc = 21, Ti = 22, Cr = 24)
[Sc(H2O)(NH3)3]3+
[Ti(en)2(NH3)2]4+
[Cr(NH3)6]3+
[Cr(NH3)6]3+
Out of TiF62-, CoF63-, Cu2Cl2 and NiCl42- (Z of Ti = 22, Co= 27, Cu = 29, Ni =28) the colourless species are
TiF62- and CoF63-
Cu2Cl2 and NiCl42-
TiF62- and Cu2Cl2
TiF62- and Cu2Cl2
Which of the following compound will exhibit cis-trans (geometrical) isomerism?
2-butene
Butanol
2-butyne
2-butyne
On the basis of the following Eo values; the strongest oxidising agent is
[Fe(CN)6]4- → [Fe(CN)6]3-] +e- ;
Eo = -0.35 V
Fe2+ → Fe3+ +e-; E = -0.77 V
[Fe(CN)6]4-
Fe2+
Fe3+
Fe3+
Which of the following complexes exhibits the highest paramagnetic behaviour?
Where gly = glycine, en = ethylenediamine and bpy = bipyridyl moities
(At. no. ; Ti = 22, V = 23, Fe =26, Co = 27)
[V(gly)2 (OH)2 (NH3)2]+
[Fe(en)(bpy)(NH3)2]+
[CO(OX)2(OH)2]-
[CO(OX)2(OH)2]-
In which of the following coordination entities the magnitude of Δo (CFSE in the octahedral field) will be maximum?
(Atomic number Co = 27)
[Co(H2O)6]3+
[CO(NH3)6]3+
[CO(CN)6]3-
[CO(CN)6]3-
Which of the following will give a pair of enantiomorphs?
(en = NH2CH2CH2NH2)
[Co(NH3)Cl2]NO2
[Cr(NH3)6]Co(CN)6]
[Co(en)2Cl2]Cl
[Co(en)2Cl2]Cl
C.
[Co(en)2Cl2]Cl
Enantiomorphs or Enantiomers: A Pair of molecules related to each other as an object to its mirror image are known as enantiomorphs or enantiomers. These are not superimposable on its mirror image.
The example is [Co(en)2Cl2]+
Dichlorobis [etylene diamine] cobalt (III)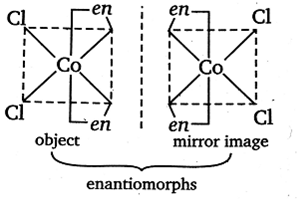
If there is no rotation of plane polarises light by a compound in a specific solvent, thought to by chiral, it may mean that:
the compound is certainly a chiral
the compound is certainly meso
there is no compound in the solvent
there is no compound in the solvent
