 Multiple Choice Questions
Multiple Choice QuestionspKa of a weak acid (HA) and pKb of a weak base (BOH) are 3.2 and 3.4, respectively. The pH of their salt (AB) solution is
7.2
6.9
7.0
7.0
In the following reactions, ZnO is respectively acting as a/an :
(a) ZnO + Na2O → Na2ZnO2
(b) ZnO + CO2 → ZnCO3
base and acid
base and base
acid and acid
acid and acid
The equilibrium constants KP1 and KP2 for the reactions X ⇌2Y and Z ⇌ P + Q, respectively are in the ratio of 1 : 9. If the degree of dissociation of X and Z be equal then the ratio of total pressure at these equilibria is
1:1
1:36
1:9
1:9
For the following three reactions a, b and c, equilibrium constants are given
a. CO (g) + H2O (g) ⇌ CO2 (g)+ H2 (g); K1
b. CH4(g) + H2O (g) ⇌ CO (g) + 3H2 (g); K2
c. CH4(g) + 2H2O (g) ⇌ CO2 (g) + 4H2 (g); K2
Which of the following relations is correct?
K1√K2 = K3
K2K3 = K1
K3 = K1K2
K3 = K1K2
Four species are listed below
i. HCO3−
ii. H3O+
iii. HSO4−
iv. HSO3F
Which one of the following is the correct sequence of their acid strength?
iv < ii < iii < i
ii < iii < i < iv
i < iii < ii < iv
i < iii < ii < iv
The pKa of a weak acid, HA, is 4.80. The pKb of a weak base, BOH, is 4.78. The pH of an aqueous solution of the corresponding salt, BA, will be
9.58
4.79
7.01
7.01
The pKa of a weak acid (HA) is 4.5. The pOH of an aqueous buffered solution of HA in which 50% of the acid is ionized is
4.5
2.5
9.5
9.5
C.
9.5
For buffer solution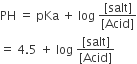
as HA is 50% ionized so [Salt] = [Acid]
pH = 4.5 pH + pOH = 14
⇒ pOH = 14 – 4.5 = 9.5
Regular use of which of the following fertilizers increases the acidity of soil ?
Potassium nitrate
Urea
Superphosphate of lime
Superphosphate of lime
An ideal gas is allowed to expand both reversibly and irreversibly in an isolated system. If Ti is the initial temperature and Tf is the final temperature, which of the following statements is correct?
(Tf)irrev > (Tf)rev
Tf > Ti for reversible process but Tf = Ti for irreversible process
(Tf)rev = (Tf)irrev
(Tf)rev = (Tf)irrev
The equilibrium constant for the reaction
is Kc = 4.9 × 10–2. The value of Kc for the reaction'
2SO2 (g) +O2(g) ⇌ 2SO3 (g) will be
416
2.40 × 10–3
9.8 × 10–2
9.8 × 10–2
