 Multiple Choice Questions
Multiple Choice Questionssp3d hybridization results in :
a square planar molecule
an octahedron molecule
a trigonal bipyramidal molecule
a tetrahedron molecule
Which of the following combination will form an electrovalent bond ?
P and Cl
NH3 and BF3
H and Ca
H and S
Which one of the following compounds has the smallest bond angle in its molecule ?
SO2
OH2
SH2
NH3
The molecule which does not exhibit dipole moment is:
NH3
CHCl3
H2O
CCl4
D.
CCl4
CCl4 does not exhibit dipole moment due to its symmetrical structure.
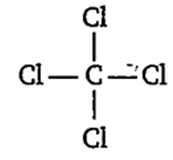
All bond angles are exactly equal to 109° 28' in:
methyl chloride
iodoform
chloroform
carbon tetrachloride
The molecular shapes of SF4, CF4 and XeF4 are
different with 1, 0 and 2 lone pairs of electrons on the central atom, respectively
different with 0, 1 and 2 lone pairs of electrons on the central atom, respectively
the same with 1, 1 and 1 lone pair of electrons on the central atoms, respectively
the same with 2, 0 and 1 lone pairs of electrons on the central atom, respectively
