 Multiple Choice Questions
Multiple Choice QuestionsWhich of the following compound possesses the C-H bond with the lowest bond dissociation energy?
Toluene
Benzene
n-pentane
2, 2-dimethyl propane
In the presence of HCl, H2S results the precipitation of group-2 elements but no group-4 elements during qualitative analysis. It is due to
higher concentration of S2-
higher concentration of H+
Lower concentration of S2-
Lower concentration of H+
Which of the following is most likely to show optical isomerism ?
HC≡C--C≡CH
HC≡C--CH3
HC≡C--H
HC≡C-C (Cl) = CH2
Acetylene reacts with HCN in the presence of Ba(CN)2 to yield :
1, 1-dicyanoethane
1, 2-dicyanoethane
vinyl cyanide
None of the above
The following compound will undergo electrophilic substitution more readily than benzene:
nitrobenzene
benzoic acid
benzaldehyde
phenol
The correct statement about the compounds (a),(b)and (c) is :
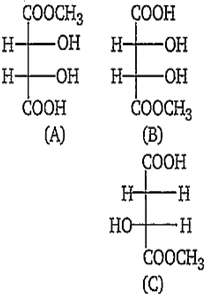
(A) and (B) are identical
(A) and (B) are diasteromers
(A) and (B) are not enantiomers
(A) and (B) are enantiomers
