 Multiple Choice Questions
Multiple Choice QuestionsHow many monochloro structural isomers are expected in free radical monochlorination of 2-methylbutane?
3
4
5
6
The correct IUPAC name of the following compound is
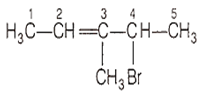
4-bromo-3-methylpent-2-ene
2-bromo-3-methylpent-4-ene
3-methyl- 4-bromopent-2-ene
3-methyl-2-bromopent-4-ene
Which one among the following cannot exhibit enantiomerism?
Diphenyl methanol
1-bromo-2-chlorobutane
2-butanol
Tartaric acid
The total number of acyclic structural isomers possible for compound with molecular formula C4H10O is
9
7
5
6
An organic compound A containing nitrogen, on acid catalysed hydrolysis produces a water soluble organic compound B and a gaseous compound C. When methyl magnesium bromide is slowly added to A in 1: 1 ratio and hydrolysed, it produces a compound which can be obtained by dry distillation of the calcium salt of B. The compound A is
N-methylmethanamide
N-ethylmethanamide
acetonitrile
N, N-dimethylmethanamide
In the estimation of sulphur by Carius method, 0.480 g of an organic compound gives 0.699 g of barium sulphate. The percentage of sulphur in this compound is (Atomic masses Ba = 137, S = 32, O = 16)
20%
15%
35%
30%
Which one of the following reactions proceeds through free radical chain mechanism?
Addition of HBr on ethene
Halogenation of benzene in the presence of FeBr3
Photochemical chlorination of methane
Hydrolysis of tert-butyl chloride with aqueous KOH
Which one of the following is the correct statement?
Achiral molecules are superimposable
Alanine is optically inactive amino acid
Glycine is optically active amino acid
There is inversion when (-)-2-methylbutan-1-ol is heated with conc. HCl to form (+)-1-chloro-2-methylbutane
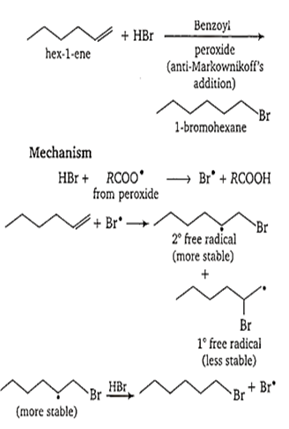
When HBr adds on hex-1-ene in the presence of benzoyl peroxide, the product is
2-bromohexane
2, 3-dibromohexane
1-bromohexane
2, 4-dibromohexane
C.
1-bromohexane
When HBr adds on hex-1-ene in the presence of benzoyl peroxide, the product formed is 1-bromohexane. The reaction is as follows-
The decreasing order of boiling points of alkyl halides is
RF > RCl > RBr > RI
RBr > RCl > RI > RF
RI > RBr > RCl > RF
RCl > RF > RI > RBr
