 Multiple Choice Questions
Multiple Choice QuestionsAssertion (A) : NH2 group of aniline is ortho, para directing in electrophilic substitutions.
Reason (R) : -NH2 group stabilises the arenium ion formed by the ortho, para attack of the electrophile.
The correct answer is
Both (A) and (R) are correct, (R) is the correct explanation of (A).
Both (A) and (R) are correct, (R) is not the correct explanation of (A).
(A) is correct, but (R) is not correct.
(A) is not correct, but (R) is correct.
In Dumas method, 0.3 g of an organic compound gave 45 mL of nitrogen at STP. The percentage of nitrogen is
16.9
18.7
23.2
29.6
The IUPAC name of the following compound is-
(CH3)2CH-CH=CH-CH=CH(C2H5)-CH3
2, 7-dimethyl-3, 5-nonadiene
2, 7-dimethyl-2-ethylheptadene
2-methyl-7-ethyl-3, 5-octadiene
1, 1-dimethyl-6-ethyl-2, 4-heptadiene
Ferric ion forms a Prussian blue coloured precipitate due to formation of
K4[Fe(CN)6]
Fe4[Fe(CN)6]3
Fe(CNS)3
K3[Fe(CN)6]
Which of the following will show geometrical isomerism?
1-butene
2-butene
2-methyl propene
Propene
n-propyl alcohol and iso-propyl alcohol are examples of
position isomerism
chain isomerism
tautomerism
geometrical isomerism
Optical isomerism is shown by
butanol-1
butanol-2
butene-1
butene-2
B.
butanol-2
The necessary and sufficient conditions for a molecule to show optical isomerism are:
(i) Compound must contain atleast one asymmetric carbon atom.
(ii) Compound has no plane of symmetry.
(iii) Compound should be non-superimposaale on its mirror image.
Butanol-2 shows optical isomerism due to presence of one asymmetric carbon atom.
The IUPAC name of the following compound is:
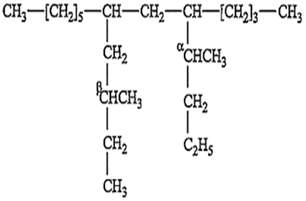
7 [β-methyl butyl]; 9 butyl tridecane
3 [β-ethyl butyl]; 9 ethyl tridecane
2 [β-ethyl ethenyl]; 8 propyl decane
None of the above
