Multiple Choice Questions
Multiple Choice QuestionsVolume of CO3 obtained by the complete decomposition of 9.85 g BaCO3 is
2.24 L
1.12 L
0.84 L
0.56 L
The reduction potential of three metallic ions X, Y and Z correspondingly are +0.52,-303 and -1.18 V. The sequence of reducing capacity of these metals will be
Y >Z >X
X >Y >Z
X >Z >Y
Z >X >Y
For the reaction . Which of the following is correct?
| Mn2+ | CO2 | H+ | |
| 5 | 2 | 4 | 10 |
| Mn+ | CO2 | H+ | |
| 2 | 5 | 10 | 16 |
| Mn2+ | C2O | CO2 | H+ |
| 6 | 8 | 16 | 18 |
| Mn2+ | C2O | CO2 | H+ |
| 10 | 12 | 24 | 12 |
Acidic K2Cr2O7 reacting with H2S2 the oxidation number of chromium is changed
from +3 to +6
from+6 to +3
from +6 to +2
remains unchanged
The process of oxidation involve
loss of electron
gain of electron
loss of proton
loss of neutron
The number of electrons transferred when KMnO4 acts as an oxidising agent to give MnO2 and Mn2+ respectively are
2, 3
1, 5
3, 5
1, 3
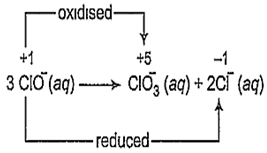
The reaction,
3ClO- (aq) → ClO (aq) + 2Cl- (aq) is an example of
oxidation reaction
reduction reaction
disproportionation reaction
decomposition reaction
C.
disproportionation reaction
A reaction in which the same species is simultaneously oxidised as well as reduced is called a disproportionation reaction. For such redox reactions to occur, the reacting species must contain an element which has atleast three oxidation states. The element in the reacting species is present in the intermediate oxidation state while the higher and lower oxidation states are available for reduction and oxidation to occur.