 Multiple Choice Questions
Multiple Choice QuestionsThe conversion of O-acylated phenol in presence of AlCl3 to C-acylated phenol is an example for this type of organic reaction
addition reaction
substitution reaction
molecular rearrangement
elimination reaction
Consider the following reaction-
C2H5Cl + AgCN X (major)
Which one of the following statements is true for X ?
(I) It gives propionic acid on hydrolysis.
(II) It has an ester functional group.
(III) It has a nitrogen linked to ethyl carbon.
(IV) It has a cyanide group.
IV
III
II
I
The order of reactivity of phenol (I), nitrobenzene (II) and benzene (III) towards nitration is
(III) > (I) > (II)
(II) > (III) > (I)
(I) > (III) > (II)
(I) > (II) > (III)
The products formed, in the reaction of phenol with Br2 dissolved in CS2 at 0°C are
o-bromo, m-bromo and p-bromophenols
o-bromo and p-bromophenols
2,4,6-tribromo and 2,3,6-tribromophenols
2,4-dibromo and 2,6-dibromophenols
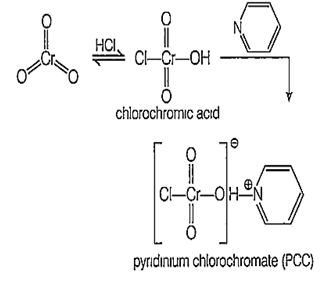
The structure of PCC is
C6H5HCrO2
C6H5HCrO2
C5H5HCrO2
C5H5HCrO3
D.
C5H5HCrO3
Pyridinium chlorochromate or PCC can be prepared by the dissolution of chromium trioxide in aqueous HCl. Addition of pyridine gives pyridinium chlorochromate as orange cryotol. PCC offers to the advantage of the selective oxidation of alcohols to aldehydes, whereas many other reagents are less selective.
C2H5OH + 4I2 + 3Na2CO3 → X + HCOONa + 5NaI + 3CO2 + 2H2O
In the above reaction, 'X' is
diodomethane
triodomethane
iodomethane
tetrariodomethane
Identify Y and Z inthe above reaction.
| X | Y |
| C6H5OH | H3CCH3 |
| X | Y |
| C2H5I | C6H5CHO |
| X | Y |
| C6H5I | H3CCH2OH |
| X | Y |
| C6H5OH | H3CCH2I |
