 Multiple Choice Questions
Multiple Choice QuestionsFor the preparation of alkanes, aqueous solution of sodium of potassium salt of carboxylic acid is subjected to
hydrolysis
electrolysis
hydrogenation
oxidation
Aldehydes can be oxidised by:
Tollen's reagent
Fehling's solution
Benedict solution
All of these
When CH2=CH-COOH is reduced with LiAlH4, the compound obtained will be
CH3-CH2-COOH
CH2=CH-CH2OH
CH3-CH2-CH2OH
CH3-CH2-CHO
Consider the acidity of the carboxylic acids
(i) PhCOOH
(ii) o-NO2C6H4COOH
(iii) p-NO2C6H4COOH
(iv) m-NO2C6H4COOH
Which of the following order is correct?
(i) > (ii) > (iii) > (iv)
(ii) > (iv) > (iii) > (i)
(ii) > (iv) > (i) > (iii)
(ii) > (iii) > (iv) > (i)
D.
(ii) > (iii) > (iv) > (i)
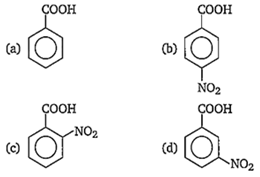
-NO2 group at any position shows electron withdrawing effect, thus acid strength is increased. But o-nitro benzoate ion is stabilised by intramolecular H-bonding like forces, hence its acid strength is maximum.
Thus, the order of acid strength is (II) > (III) > (IV) > (I).
In a set of the given reactions, acetic acid yielded a product C.
CH3COOH + PCl5 → A
A C
Product C would be
CH3CH(OH)C6H5
CH3-(OH)C6H5
CH3CH(OH)C2H5
CH3COC6H5
Isomers of propionic acid are
HCOOC2H5 and CH3COOCH3
HCOOC2H5 and C3H7COOH
CH3COOCH3 and C3H7OH
C3H7OH and CH3COCH3
Metamers of ethyl propionate are
C4H9COOH and HCOOC4H9
C4H9COOH and CH3COOC3H7
CH3COOCH3 and CH3COOC3H7
CH3COOC3H7 and C3H7COOCH3
What is the correct IUPAC name of

4-methoxy-2-nitrobenzaldehyde
4-formyl-3-nitro anisole
4-methoxy-6-nitrobenzaldehyde
2-formyl-5-methoxy nitrobenzene
2-pentanone and 3-pentanone can be distinguished by :
Cannizaro's reaction
Aldol condesation
Iodoform reaction
Clemmensen's reduction
The acid which contains the aldehyde group is:
acetic acid
formic acid
benzoic acid
propionic acid
