 Multiple Choice Questions
Multiple Choice QuestionsThe molecule having zero dipole moment is
CH2Cl2
BF3
NF3
ClF3
B.
BF3
The dipole moment of BF3 molecule is zero due to its symmetrical (triangular planar) structure. The three fluorine atoms lie at the corners of an equilateral triangle with boron at the centre. Thus, the vectorial addition of the dipole moments of the three bonds gives a netsum of zero.
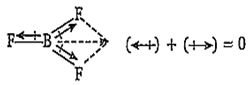
The correct order of decreasing dipole moment of (I) toluene, (II) m-dichlorobenzene (III) o-dichlorobenzene and (IV) p-dichlorobenzene is
IV < II < I < III
IV < I < II < III
I < IV < II < III
IV < I < III < II
Among the following compounds of boron, the species which also forms -bond in addition to σ-bonds is
BH3
B2H6
BF3
BF
Match the following
| List I | List II |
| (a) PCl3 | (i) Square planar |
| (b) BF3 | (ii) T-shape |
| (c) ClF3 | (iii) Trigonal pyramidal |
| (d) XeF4 | (iv) See- saw |
| (v) Trigaonal planar |
(a) (b) (c) (d)
(iv) (ii) (i) (iii)
(a) (b) (c) (d)
(iii) (v) (ii) (iv)
(a) (b) (c) (d)
(iii) (v) (ii) (i)
(a) (b) (c) (d)
(iii) (v) (ii) (v)
