 Multiple Choice Questions
Multiple Choice QuestionsWhich of the following is the wrong statement?
ONCl and ONO- are not isoelectronic
O3 molecule is linear
Ozone is violet-black in solid state
Ozone is violet-black in solid state
Which of the following statement is wrong?
The stability of hydrides increases from NH3 to BiH3 in group 15 of the periodic table.
Nitrogen cannot form dπ-pπ bond.
Single N - N bond is weaker than the single P - P bond.
Single N - N bond is weaker than the single P - P bond.
Which one of the following orders presents the correct sequence of the increasing basic nature of the given oxides?
Al2O3 < MgO < Na2O < K2O
MgO < K2O < Al2O3 < Na2O
Na2O < K2O < MgO < Al2O3
Na2O < K2O < MgO < Al2O3
Which of the following statements regarding sulphur is incorrect?
S2 molecule is paramagnetic.
The vapour at 200ºC consists mostly of S8 rings.
At 600ºC the gas mainly consists of S2 molecules.
At 600ºC the gas mainly consists of S2 molecules.
The structure of IF7 is
square pyramid
trigonal bipyramid
octahedral
octahedral
D.
octahedral
The structure is pentagonal bipyramid having sp3d3 hybridisation as given below,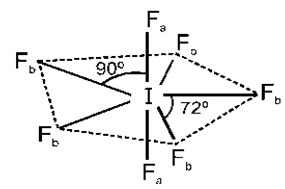
29.5 mg of an organic compound containing nitrogen was digested according to Kjeldahl’s method and the evolved ammonia was absorbed in 20 mL of 0.1 M HCl solution. The excess of the acid required 15 mL of 0.1 M NaOH solution for complete neutralization. The percentage of nitrogen in the compound is
59.0
47.4
23.7
23.7
Which of the following reactions is an example of a redox reaction?
XeF4 + O2F2 → XeF6 + O2
XeF2 + PF5 → [XeF]+PF6–
XeF6 + H2O → XeOF4 + 2HF
XeF6 + H2O → XeOF4 + 2HF
The products obtained when chlorine gas reacts with cold and dilute aqueous NaOH are
ClO– and ClO3–
ClO2- and ClO3-
Cl– and ClO–
Cl– and ClO–
