 Multiple Choice Questions
Multiple Choice QuestionsCH3C(CH2CH3) = CH(CH3) X. No. of optical isomer possible will be
2
4
0
3
A.
2
When A reacts with hydrogen in presence of a catalyst the following reaction is observed,
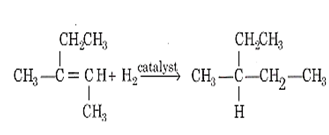
The product formed is in both d and l form. Therefore, two optical isomers are possible.
Which of the following is the correct increasing order of stability of free radicals.
(CH3)2CH0 < (CH3)2CH0 < (C6H5)20CH < (C6H5)3C0
(CH3)2CH0 < (CH3)2C0 < (C6H5)30C < (C6H5)2C0
(C6H5)30C < (C6H5)20CH < (CH3)30C < (CH3)20CH
(C6H5)20CH < (C6H5)2C0 < (CH3)30C < (CH3)20CH
The number of enantiomers and mesa forms for the compound ,
HOOC-CH(CH3)-CHOH-CHBr-CHOHCH(CH3)-COOH are respectively:
12 , 2
12 , 4
8 , 4
8 , 2
Which of the following yields as a major product is mono chlorination of ethyl benzene in the presence of light and heat.
m- chlorotoluene
a mixture of -O- and p-chlorotoluene
1-chloro-2- phenylethane
1-chloro -1- phenyl ethane
The correct IUPAC name of

4-methyoxy -2- nitrobenzaldehyde
4-formyl -3- nitro anisole
4- methoxy -6- nitrobenzaldehyde
2-formyl -5- methoxy nitrobenzene
The correct order of stability of carbocations, is :
1 < 2 < 3 < CH3
H3 < 1 < 2 < 3
3 < 2 < 1 < H3
2 < 3 < 1 < H3
The IUPAC name of the following compound is

bicyclo [2, 2, 0] octane
bicyclo [0, 2, 2] hexane
bicyclo [2, 1, 1] hexane
bicyclo [2, 2, 0] hexane
The following compound can not exhibit
CH3CH = CH - CH(OH).COOH
geometrical isomerism
optical isomerism
metamerism
position isomerism
