 Multiple Choice Questions
Multiple Choice QuestionsConsider the reactions :
A-Methoxymethane, X-Ethanoic acid, Y-Acetate ion, Z-hydrazine
A-Methoxymethane, X-Ethanol, Y-Ethanoic acid, Z-Semicarbazide
A-Ethanal, X-Ethanol, Y-But-2-enal, Z-Semicarbazone
A-Ethanal, X-Ethanol, Y-But-2-enal, Z-Semicarbazone
The IUPAC name of the compound
3-keto-2-methylhex-4-enal
5-formylhex-2-en-3-one
5-methyl-4-oxohex-2-en-5-al
5-methyl-4-oxohex-2-en-5-al
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their
Formation of intramolecular H-bonding
Formation of intermolecular H-bonding
Formation of intermolecular H-bonding
More extensive association of carboxylic acid via Vander Waals force of attraction
Compound A, C8H10O, is found to react with NaOI (produced by reacting Y with NaOH) and yields a yellow precipitate with characteristic smell. A and Y are respectively

![]()


D.

Haloform reaction is shown by compound having

The product of acid hydrolysis of (P) and (Q) can be distinguished by
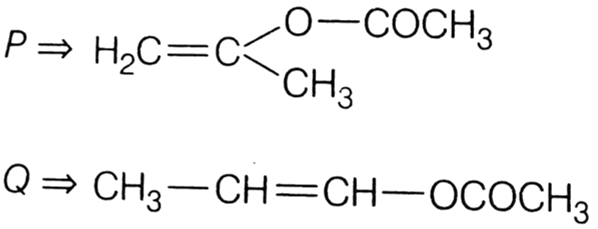
Lucas reagent
2,4-DNP
Fehling's solution
NaHSO3
Clemmensen reaction of the ketone is carried out in the presence of
LiAlH4
Zn-Hg with HCl
glycol with KOH
H2 with Pt as a catalyst
Consider the following compounds
Cl3C - COOH Br3C - COOH I3C - COOH
(I) (II) (III)
The decreasing order of decarboxylation is
I > II > III
III > II > I
III > I > II
II > I > III
