 Multiple Choice Questions
Multiple Choice QuestionsAssertion : CH3OCH3 and C2H5OH has comparable molecular weight but boiling point of C2H5OH is more than dimethyl ether.
Reason : C2H5OH forms intermolecular Hbonding while CH3OCH3 forms intramolecular H-bonding.
If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
If Assertion is true but Reason is false.
If Assertion is false but Reason is true.
Upon treatment with I2 and aqueous NaOH, which of the following compounds will form iodoform?
CH3CH2CH2CH2CHO
CH3CH2COCH2CH3
CH3CH2CH2CH2CH2OH
CH3CH2CH2CH(OH)CH3
Upon treatment with Al(OEt)3 followed by usual reactions (work up), CH3CHO will produce:
only CH3COOCH2CH3
a mixture of CH3COOH and EtOH
only CH3COOH
only EtOH
The most contributing tautomeric enol form of MeCOCH2CO2 Et is:
CH2=C(OH)CH2CO2Et
MeC(OH)=CHCO2Et
MeCOCH=C(OH)OEt
CH2=C(OH)CH=C(OH)OEt
By passing excess Cl2(g) in boiling toluene, which one of the following compounds is exclusively formed?



![]()
D.
![]()

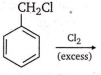

![]()
An equimolar mixture of toluene and chlorobenzene is treated with a mixture of conc. H2SO4 and conc. HNO3. Indicate the correct statement from the following.
p-nitrotoluene is formed in excess
equimolar amounts of p-nitrotoluene and p-nitro chlorobenzene are formed
p-nitro chlorobenzene is formed in excess
m-nitro chlorobenzene is formed in excess
Among the following compounds, the one(s) that gives (give) effervescence with aqueous NaHCO3 solution is (are)
I and II
I and III
Only II
I and IV
 Long Answer Type
Long Answer TypeAn organic compound with molecular formula C9H10O forms 2, 4-DNP derivative, reduces Tollen's reagent and undergoes Cannizaro reaction. On vigorous oxidation, it gives a dicarboxylic acid which is used in the preparation of terylene. Identify the organic compound.
Deep blue CuSO4.5H2O is converted to a bluish-white salt at 100oC. At 250oC and 750oC it is then transformed to white powder and black material respectively. Identify the salts.
