 Multiple Choice Questions
Multiple Choice QuestionsA compound is soluble in conc. H2SO4 . It does not decolourise bromine in CCL4 but in oxidised by chromic anhydride in aqueous H2SO4 with 2s , turning orange solution to blue green , afterward opaque. The original solution contains :
a primary alcohol
a secondary alcohol
a tertiary alcohol
an ether
Which of following is the correct order of increasing reactivity?
RCOOR' < RCOCl < RCONH2
RCOOR' < RCONH2 < RCOCl
RCOCl < RCONH2 < RCOOR'
RCONH2 < RCOOR' < RCOCl
What is the name of reaction when benzaldehyde changes in to benzyl alcohol?
Cannizzaro's reaction
Wurtz reaction
Kolbe reaction
Friedel-Craft's reaction
A sample of CHCl3 before using as an anaesthetic is tested by
AgNO3 solution after boiling with KOH
Ammonical cuprous chloride
Fehling solution
AgNO3
What is the correct order of acidity from weakest to strongest acid for these compounds :
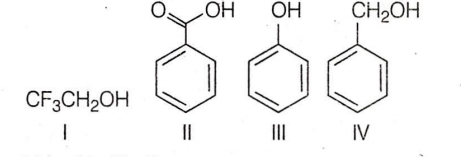
I < IV < III < II
III < IV < III < II
IV < I < III < II
II < III < I < IV
D.
II < III < I < IV
Benzoic acid is more acidic than phenol , because it is more resonance stabilised and the negative charge on the carboxylate ion is delocalised over two oxygen atoms.
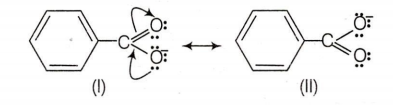
Phenol is more acidic than CF3CH2OH, because of its resonance stabilised phenoxide ion. Further more, CF3CH2OH is more acidic as compared to benzyl alcohol, because of the presence of more electron withdrawing groups. Thus, the correct order of acidity is :

Lucas test is given by an alcohol within 5 minutes. 0.22 g of which liberates 56 mL of CH4 at STP on treating with CH3MgI. The structure of alcohol is
CH3 - CHOH - CH3
(CH3)2CH - CHOH - CH3
CH3 - CH2 - CHOH - CH3
(CH3)3C - CHOH - CH3
The reaction in which phenol differs from alcohol is :
it undergoes esterification with carboxylic acid
it reacts with ammonia
it forms yellow crystals of iodoform
it liberates H2 with Na metal
