 Short Answer Type
Short Answer TypeCorrect the following statements:
For example: chlorine is a bleaching agent.
Should read: moist chlorine is a bleaching agent
ô Lead bromide conducts electricity.
An aqueous solution of Nickel sulphate contains Ni2+ and SO2-3 ions.
i) which ion moves towards the cathode?ô
ii) what is the product at the anode?ô
Find the odd one out and explain your (note: valency is not a criterion)
Copper, lead, zinc, mercury.
Correct the following statement:
For example: chlorine is a bleaching agent.
Should read : moist chlorine is a bleaching agent.
Haematite is the chief ore of aluminium.
The sketch below illustrates the refining of aluminium by Hoopeãs process.
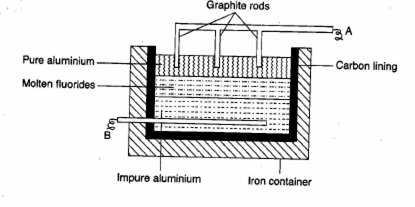
(I) Which of A and B is the cathode and which one is the anode?ô
(II) What is the electrolyte in the tank?ô
(III) What material is used for the cathode?ô
I) A is cathode and Bô is an anode.
II) Moltenô fluorides.
III)ô Graphiteô rodsô dipped in pure molten aluminium.
State the property of metal being utilised in the following:
|
Use of metal |
Property |
|
Zinc in Galvanization |
ô |
|
Aluminium in Thermite welding |
ô |
Write a fully balanced equation for each of the following cases:
Red leads is warmed with concentrated hydrochloric acid
Write a fully balanced equation for each of the following cases:
Magnesium metal is treated with dilute hydrochloric acid.
Correct the following statement:
For example : chlorine is a bleaching agent
Should read: moist chlorine is a bleaching agent.
Hydrochloric acid is prepared in the laboratory by passing hydrogen chloride directly through the water.
Write a fully balanced equation for each of the following cases:
Magnesium nitrides is treated with warm water.
