 Short Answer Type
Short Answer TypeAnswer the following questions:
How will you distinguish between Ammonium hydroxide and Sodium hydroxide using copper sulphate solution?
How will you distinguish between dilute hydrochloric acid and dilute sulphuric acid using lead nitrate solution?
Identify the salts P and Q from the observations given below:
i. On performing the flame test salt P produces a lilac coloured flame and its solution gives a white precipitate with silver nitrate solution, which is soluble in Ammonium hydroxide solution.
ii. When dilute HCl is added to a salt Q, a brisk effervescence is produced and the gas turns lime water milky. When NH4OH solution is added to the above mixture (after adding dilute HCl), it produces a white precipitate which is soluble in excess NH4OH solution.
Draw an electron dot diagram to show the formation of each of the following compounds:
Methane
Electron dot structure of Methane:-
Electronic configuration of Carbon 2,4. Carbon requires four more electrons to stable itself. Therefore, one atom of carbon shares four electron pairs, one with each of the four atoms of hydrogen.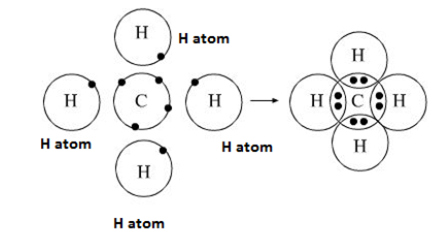
Draw an electron dot diagram to show the formation of each of the following compounds:
Magnesium Chloride
[Mg = 12, Cl = 17]
State the observations at the anode and at the cathode during the electrolysis of:
fused lead bromide using graphite electrodes.
State the observations at the anode and at the cathode during the electrolysis of:
copper sulphate solution using copper electrodes.
Select the ion in each case, that would get selectively discharged from the aqueous mixture of the ions listed below:
i. SO42-, NO3- and OH-
ii. Pb2+, Ag+ and Cu2+
|
Lab |
Reactants used |
Products formed |
Drying Agent |
Method of |
|
| (i) | HCl gas | NaCl + H2SO4 | A | conc. H2SO4 | B |
| (ii) | NH3 gas | C |
Mg(OH)2 |
D | E |
Write balanced chemical equations to show:
The oxidizing action of conc. Sulphuric acid on carbon.
