 Short Answer Type
Short Answer TypeWrite balanced chemical equations to show:
The behavior of H2SO4 as an acid when it reacts with Magnesium.
Write balanced chemical equations to show:
The dehydrating property of conc. Sulphuric acid with sugar.
Write balanced chemical equations to show how SO3 is converted to Sulphuric acid in the contact process.
Propane burns in air according to the following equation,
C3H8 + 5O2 → 3CO2 + 4H2O
What volume of propane is consumed on using 1000 cm3 of air, considering only 20% of air contains oxygen?
Given:
C3H8 + 5O2 → 3CO2 + 4H2O
Volume of air = 1000 cm3
Percentage of oxygen in air = 20%
From the given information,
C3H8 + 5O2 → 3CO2 + 4H2O
1vol 5vols 3vols 4 vols
According to Gay-Lussac’s law,
1 vol. of propane consumes 5 vol. of oxygen.
Volume of oxygen = 1000 cm3 × 20% = 200 cm3
Therefore, Volume of propane burnt for every 200 cm3 of oxygen,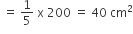
40 cm3 of propane is burnt.
The mass of 11.2 litres of a certain gas at s.t.p. is 24 g. Find the gram molecular mass of the gas.
A gas cylinder can hold 1 kg of hydrogen at room temperature and pressure.
Find the number of moles of hydrogen present.
A gas cylinder can hold 1 kg of hydrogen at room temperature and pressure
What weight of CO2 can the cylinder hold under similar conditions of temperature
and pressure? (H = 1, C = 12, O = 16)
A gas cylinder can hold 1 kg of hydrogen at room temperature and pressure
i. If the number of molecules of hydrogen in the cylinder is X, calculate the number of CO2 molecules in the cylinder under the same conditions of temperature and pressure.
ii. State the law that helped you to arrive at the above result.
Write a balanced chemical equation for the preparation of the following salts:
Copper carbonate
Write a balanced chemical equation for the preparation of the following salts:
Ammonium sulphate crystals
