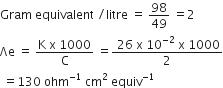Short Answer Type
Short Answer TypeA solution is prepared by dissolving 2.0 g of sucrose and 2.0 g of urea in 100 g of water at 298 K. Calculate the vapour pressure of the solution, if the vapour pressure of pure water at 298 K is 23.756 torr.
(Molecular weight of urea = 60 and sucrose = 342)
The average life-period of a radioactive nucleide is 100 hours. How long will it take for 10 g of the nucleide to lose 75 percent of its radioactivity ? [4]
(ii) Calculate the number of α -particles and β-particles emitted in a total transformation from .
.
(i) What is phase rule ? Draw the phase diagram of water and label all the parts representing the phases and curves. Mark the triple point.
(ii) Calculate the equivalent conductivity of 1 M H2SO4, whose specific conductivity is 26 x 10 – 2 ohm –1 cm–1.
(i) Phase Rule : According to phase rule, there is a definite relation in a system between the number of degrees of freedom, number of components and number of phases present.
According to this rule
F = C – P + 2
F = Number of degrees of freedom
C = Number of components
P = Number of phases
Numeral 2, represent two additional variables, temperature and pressure, besides the concentration variable.
Phase diagram of water
OP and ON represent variations of vapour-pressure with temperature of water and ice respectively.
OQ equilibrium between ice and water.
O Triple point.
At point O, all three phases ice, water and water vapour are in equilibrium.
(ii) Δe = Equivalent conductance
K = Specific conductance
Equivalent weight of H2SO4 = 49