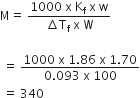Short Answer Type
Short Answer TypeCorrect the following statements:
Chloroacetic acid is more acidic than acetic acid because of the - M effect.
Correct the following statements:
When acetone is treated with bleaching powder, acetaldehyde is obtained.
Given Kb = 1.86 K Kg mole– 1, wt. of solute (water) = w = 1.70 g
Volume of water = Weight of water (∵ density of water = 1 g/cc)
Weight of solvent (water) = W = 100 g
Freezing point of solution = –0.093°C
Depression in freezing point = ΔTf
= Freezing point of water — Freezing point of solution
= 0-(-0.093)
= 0.093°C
Molecular weight of cane sugar = M = ?