 Short Answer Type
Short Answer TypeThe formation of chlorobenzene from benzene is a SN2 reaction.
Explain the following :
Zinc displaces hydrogen from acid solution [E°Zn+2/Zn =-0.76 volts].
(i) Given E0Zn2+/Zn =-0.76 V, E°H+/H =0 V
The following reaction will take place during displacement of hydrogen in an acidic solution of zinc.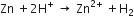
∴Zinc acts as anode and hydrogen as a cathode.
E°cell E°C - E°A
= 0 - (0.76)
= + 0.76 V
Since the E°cell is positive this reaction takes place and zinc displace hydrogen from an acidic solution of zinc.
Explain the following :
The hydride of sulphur is a gas but the hydride of oxygen is a liquid.
