 Short Answer Type
Short Answer TypeGive a reason for each of the following :
The number of hydrogen ions in an aqueous solution of acetic acid increase considerably with dilution while this is not the case with an aqueous solution of hydrogen chloride.
Give a reason for each of the following :
A mixture of NH4Cl and NH4OH is used to precipitate the metallic hydroxides of group III.
Given,
T = 27°C = 273 + 27 = 300 K,
R = 1.987 cal deg-1 mol-1, V1 = 7 L, V2 = 14 L
Mass of oxygen = 64 g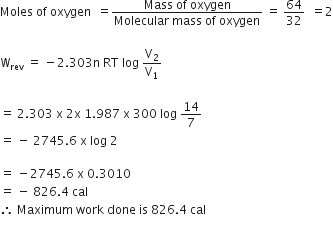
Arrange the following in increasing order of acidity and explain your order:
Phenol, Methanol, Water.
