 Short Answer Type
Short Answer TypeGive a reason for each of the following :
The number of hydrogen ions in an aqueous solution of acetic acid increase considerably with dilution while this is not the case with an aqueous solution of hydrogen chloride.
Give a reason for each of the following :
A mixture of NH4Cl and NH4OH is used to precipitate the metallic hydroxides of group III.
Arrange the following in increasing order of acidity and explain your order:
Phenol, Methanol, Water.
Silicon carbide is manufactured by heating a mixture of sand (54%), coke (34%), saw dust (10%) and salt (2%) in an electric furnace at a temperature between 2270 - 2570K.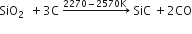
The salt acts as a flux and removes metallic impurities. Saw dust makes mass porous. The electric furnace made of fine bricks is used to manufacture SiC. The side walls of the furnace can be removed at the end of reaction to take out the product on the completion of the reaction. Each end of the furnace is provided with a carbon electrode. When heavy current is passed through furnace for 36 hours at a temperature between 2270-2570K silica combines with carbon to form silicon carbide. When the reaction is complete the side walls of the furnace are pulled out and black crystals of SiC are removed from it. These crystals are washed crushed and washed, with H2S04, NaOH and H20 one after the other. Then it is dried to get pure silica.
