 Short Answer Type
Short Answer TypeWrite balanced equations for each of the following reactions :
(i) Silver Sulphide and Sodium Cyanide.
(ii) Water is added to aluminium chloride.
(iii) Bromine water and sodium sulphite.
(iv) Phosphorous is boiled with a solution of sodium hydroxide.
How will you bring about the following conversions :
(i) Chloroacetic acid to Glycine.
(ii) Formic acid to Oxalic acid.
(iii) Diethyl ether to ethanol.
Give one example each of the following name reactions :
(i) Hofmann’s Degradation
(ii) Cannizzaro’s Reaction
(iii) Benzoin Condensation
(iv) Friedel Craft’s Reaction.
(i) Hofmann’s degradation : When primary amide is heated with bromine in presence of an alkali, a primary amine with one carbon atom less than the parent amide is obtained. For example, ethanamide gives methyl amine on reaction with bromine and sodium hydroxide.
![]()
(ii) Cannizzaro’s reaction : The reaction involving simultaneous oxidation and reduction of an aldehyde which do not have α-hydrogen atom in presence of aq. alkali is called Cannizzaro reaction. For example, formaldehyde on reaction with aq. NaOH forms methyl alcohol and sodium formate.![]()
(iii) Benzoin condensation : When aromatic aldehydes react with ale. KCN, undergo self-condensation reaction it is called benzoin condensation. For example, benzaldehyde on heating with alc. KCN forms benzoin.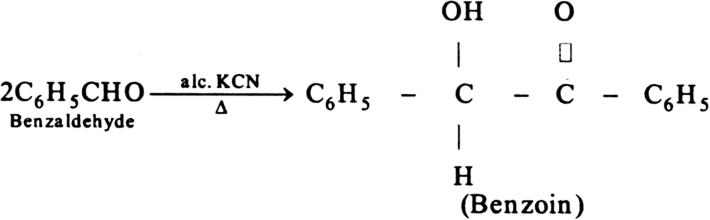
(iv) Friedel Craft’s reaction : The replacement of hydrogen atom of benzene ring by alkyl group in presence of anhydrous AlCl3 is called Friedel Craft’s reaction. For example, benzene reacts with methyl chloride in presence of anhydrous AlCl3 to form toluene.
How will you distinguish between the following pairs of compounds ? Give one good chemical test.
(i) Glucose and Sucrose
(ii) Ethanol and Phenol
Write balanced equations of the following reactions :
(i) Aniline and Bromine water
(ii) Ethylamine and Nitrous Acid
(iii) Acetic Anhydride and Ammonia
(iv) Acetone and Phosphorous Pentachloride.
 Long Answer Type
Long Answer Type