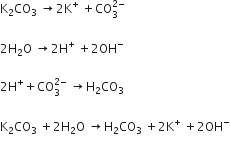Short Answer Type
Short Answer TypeAnswer the following questions:
Write the relationship between Gibb’s free energy, enthalpy, entropy and the temperature of a system. What is the equation known as?
Answer the following questions:
The elevation of boiling point produced by dilute equimolar solutions of three substances is in the order A> glucose>B. Suggest a reason for this observations.
Give reason for the following:
(i) The density of ice is less than that of water.
(ii) A solution of potassium carbonate turns red litmus paper blue while that of potassium sulphate has no effect on litmus.
i) Ice has highly ordered, three-dimensional, hydrogen bonded structure as shown below.
Each oxygen atom is tetrahedrally bonded to four other oxygen atoms at a distance of 276 pm. Hydrogen bonding gives ice a rather open type structure with wide holes. These holes can hold some other molecules of appropriate size interstitially.
(ii) An aqueous solution of K2C03 is basic in nature due "to hydrogen’s but K2S04 have a more covalent character that is why it is not hydrogen’s.