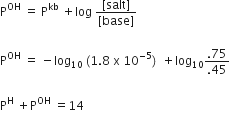Short Answer Type
Short Answer TypeAnswer the following questions:
Write the relationship between Gibb’s free energy, enthalpy, entropy and the temperature of a system. What is the equation known as?
Answer the following questions:
The elevation of boiling point produced by dilute equimolar solutions of three substances is in the order A> glucose>B. Suggest a reason for this observations.
Give reason for the following:
(i) The density of ice is less than that of water.
(ii) A solution of potassium carbonate turns red litmus paper blue while that of potassium sulphate has no effect on litmus.
i) To obtain pure NaCl solution the crude salt is dissolved in minimum amount of H20 and filtered to remove insoluble impurities. The solution is then saturated with HCl gas. Crystals of pure NaCl separated out. Ca and Mg chloride being more soluble than NaCl, remain in solution.
ii)
We have given,
[Base] = .45 mole L-1,
[salt] = .75 mole L-1
Kb= 1.8xl0-5
According to the Henderson’s equation