 Short Answer Type
Short Answer TypeAnswer the following questions:
Write the relationship between Gibb’s free energy, enthalpy, entropy and the temperature of a system. What is the equation known as?
Answer the following questions:
The elevation of boiling point produced by dilute equimolar solutions of three substances is in the order A> glucose>B. Suggest a reason for this observations.
Give reason for the following:
(i) The density of ice is less than that of water.
(ii) A solution of potassium carbonate turns red litmus paper blue while that of potassium sulphate has no effect on litmus.
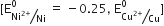
The standard Potential of nickel is less than that copper it is above copper in the Electrochemical series and possesses a greater reducing power. So Ni can displace Cu from CuSO4 solution.
Cu2+ +Ni(s) --> Cu(s) +Ni2+(aq)
