 Short Answer Type
Short Answer TypeAnswer the following questions:
Write the relationship between Gibb’s free energy, enthalpy, entropy and the temperature of a system. What is the equation known as?
Answer the following questions:
The elevation of boiling point produced by dilute equimolar solutions of three substances is in the order A> glucose>B. Suggest a reason for this observations.
Give reason for the following:
(i) The density of ice is less than that of water.
(ii) A solution of potassium carbonate turns red litmus paper blue while that of potassium sulphate has no effect on litmus.
according to Kohlraush’s law of Independent migration of ions - the molar conductivity at infinite dilution of an electrolyte is equal to the same of molar conductance term multiplied by the number of respective ion present in the formula unit of the electrolyte. If λ+ ∞and λ- ∞respective represent the molar conductivities of cations and anions of an electrolyte, then according to Kohlraush’s law.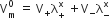
Where V+ and V_ is the Number of cations and anions.
