 Short Answer Type
Short Answer TypeWhat is the electronic configuration of chromium atom (Z = 24)? Give a reason for your answer.
i) State the second law of thermodynamics in terms of the entropy of the universe.
(ii) Calculate the maximum work that can be obtained from the given electrochemical cell constructed with two metals M and N.
The cell reaction is M + N+2 → M+2 + N
To precipitate group III cations NH4Cl should be added to the solution before the addition of ammonium hydroxide. Explain why.
Solubility products of group (III) hydroxides are low as compared to the hydroxides of higher groups. So a low concentration of OH- ions is required to precipitate group (III) cations. The presence of NH4Cl (a strong electrolyte) suppresses dissociation of weak electrolyte NH4OH by common ion effect and decreases OH-ion concentration considerably.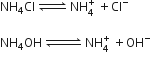
A study of chemical kinetics of the reaction A+B → products, gave the following data at 25°C:

Find:
(1) The order of reaction with respect to A.
(2) The order of reaction with respect to B.
(3) The rate law.
