 Short Answer Type
Short Answer Type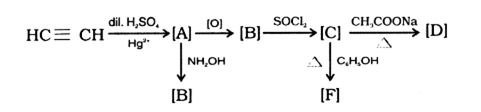
Write balanced chemical equations fro the following reactions and name the reactions:
(i) Acetamide is heated with bromine and sodium hydroxide solution.
(ii) Benzaldehyde is treated with 58% and sodium hydroxide solution.
Give one chemical test to distinguish between the following pairs of compounds:
(i) Acetone and phenol.
(ii) Formic acid and Acetic acid.
(i) Name the type of isomerism exhibited by the following pairs of compounds.
(1) (C2H5)2NH and CH3-NH-C3H7.
(2) 1-butanol and 2 methyl-1-propanol.
(ii) Name the type of isomerism that the compound with molecular formula C3H602 exhibits. Represent the isomers.
Write balanced chemical equations for the following reactions:
(i) Oxalic acid is treated with acidified potassium permanganate solution.
(ii) Benzoic acid is treated with a mixture of concentrated nitric acid and concentrated sulphuric acid.
(iii) Methyl magnesium iodide is treated with carbon dioxide and the product hydrolysed in acidic medium.
(iii) Ethylacetate is treated with ammonia.
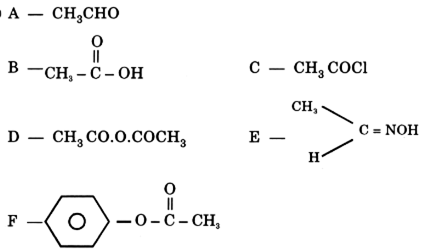
(i) Identify [A], [B], [C], [D] and [E],
(ii) Write balanced chemical equation of [D] with chlorine is the presence of red phosphorus and name the reaction. compounds relevant to these isomers.