 Short Answer Type
Short Answer TypeAnswer the following question:
Why does the density of transition elements increase from Titanium to Copper? (at. no. Ti = 22,
Cu = 29)
Calculate the osmotic pressure of a solution prepared by dissolving 0.025 g of K2SO4 in 2.0 litres of water at 25oC assuming that K2SO4 is completely dissociated. (mol. wt. of K2SO4 = 174 g mol-1)
What type of isomerism is shown by the following coordination compounds:
[PtCl2(NH3)4]Br2 and [PtBr2(NNH3)4]Cl2
Write their IUPAC names.
(i) Write the rate law expression for the reaction A+ B +C → D +E, if the order of reaction is first, second and zero with respect to A, B, and C, respectively.
(ii) How many times the rate of reaction will increase if the concentration of A, B and C are doubled in the equation given in (i) above?
The rate of reaction becomes four times when the temperature changes from 293 K to 313 K. Calculate the energy of activation (Ea) of the reaction assuming that it does not change with temperature. (R = 8.314 J K–1 mol–1)
State the role of the following chemicals in the food industry:
(i) Sodium benzoate
(ii) Aspartame
An aromatic organic compound [A] on heating with NH3 and Cu2O at high pressure gives [B]. The compound [B] on treatment with an ice-cold solution of NaNO2 and HCI gives [C], which on heating with Cu/HCl gives compound [A] again. Identify the compounds [A], [B] and [C]. Write the name of the reaction for the conversion of [B] to [C].
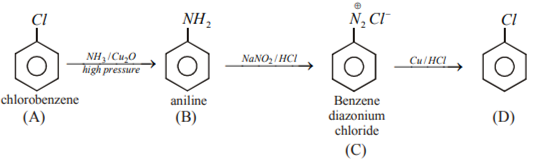
Conversion of B to C is called Sand Meyer reaction.
