 Multiple Choice Questions
Multiple Choice QuestionsEquivalent conductivity at infinite dilution for sodium-potassium oxalate [(COO-)2 Na+K+] will be [given molar conductivities of oxalate, K+ and Na+ ions at infinite dilution are 148.2, 50.1, 73.5 S cm mol' respectively]
271.8 S cm2 eq-1
67.95 S cm2 eq-1
543.6 S cm2 eq-1
135.9 S cm2 eq-1
On heating, chloric acid decomposes to
HClO4, Cl2, O2 and H2O
HClO2, Cl2, O2 and H2O
HClO, Cl2O and H2O2
HCl, HClO, Cl2O and H2O
In the following species, the one which is likely to be the intermediate during benzoin condensation of benzaldehyde is
![]()
![]()
![]()
![]()
Baeyer's reagent is
alkaline potassium permanganate
acidified potassium permanganate
neutral potassium permanganate
alkaline potassium manganate
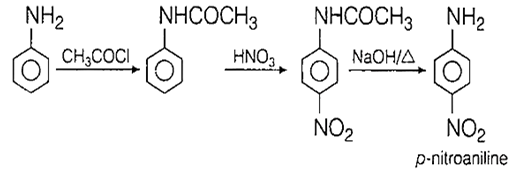
When aniline is nitrated with nitrating mixture in ice cold condition, the major product obtained is
p-nitroaniline
2,4-dinitroaniline
o-nitroaniline
m-nitroaniline
A.
p-nitroaniline
Direct nitration of aniline with nitric acid gives a complex mixture of mono, di- and tri-nitro compounds and oxidation products. If NH2 group is protected by acetylation and then nitrated with nitrating mixture, p-isomer is the main product.
Ribose and 2-deoxyribose can be differentiated by
Fehling's reagent
Tollen's reagent
Barfoed's reagent
Osazone formation
The correct order of acid strength of the following substituted phenols in water at 28°C is
p-nitrophenol < p-fluorophenol < p-chlorophenol
p-chlorophenol < p-fluorophenol < p-nitrophenol
p-fluorophenol < p-chlorophenol < p-nitrophenol
p-flurophenol < p-nitrophenol < p-chlorophenol
The best method for preparation of Me3CCN is
to react with Me3COH with HCN
to react with Me3CBr with NaCN
to react with Me3CMgBr with ClCN
to react Me3CLi with NH2CN
Correct statement(s) in cases of n-butanol and t-butanol is (are)
both are having equal solubility in water
t-butanol is more soluble in water than n-butanol
boiling point of t-butanol is lower than n-butanol
boiling point of n-butanol is lower than t-butanol